Protons Neutrons And Electrons Chart
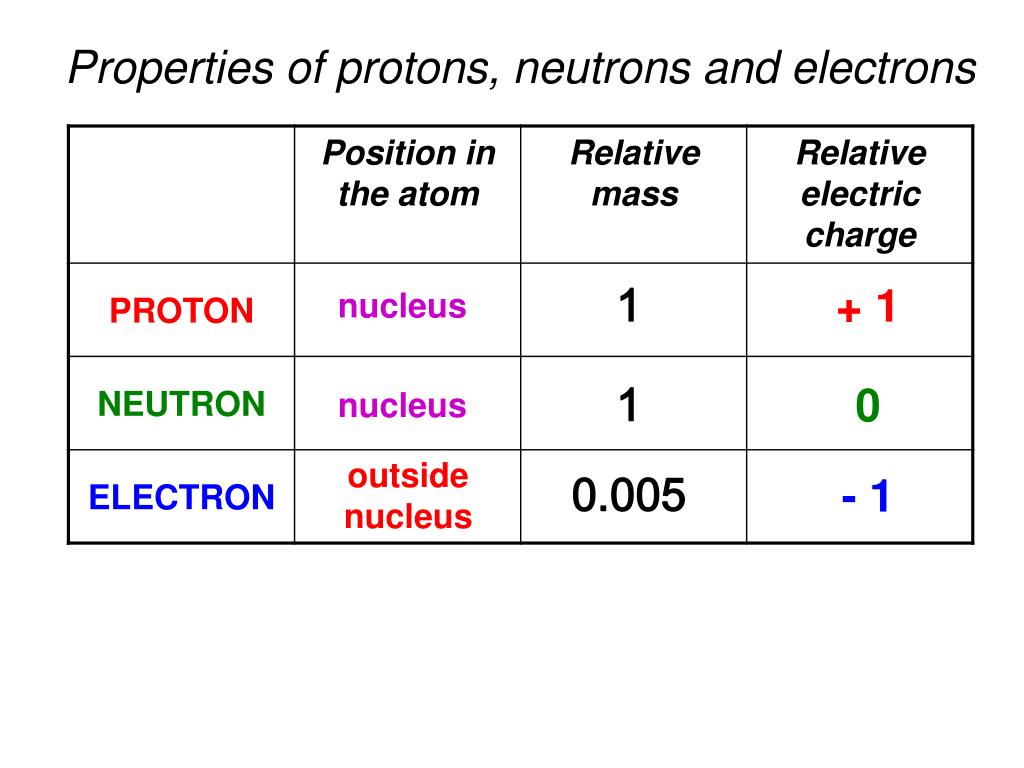
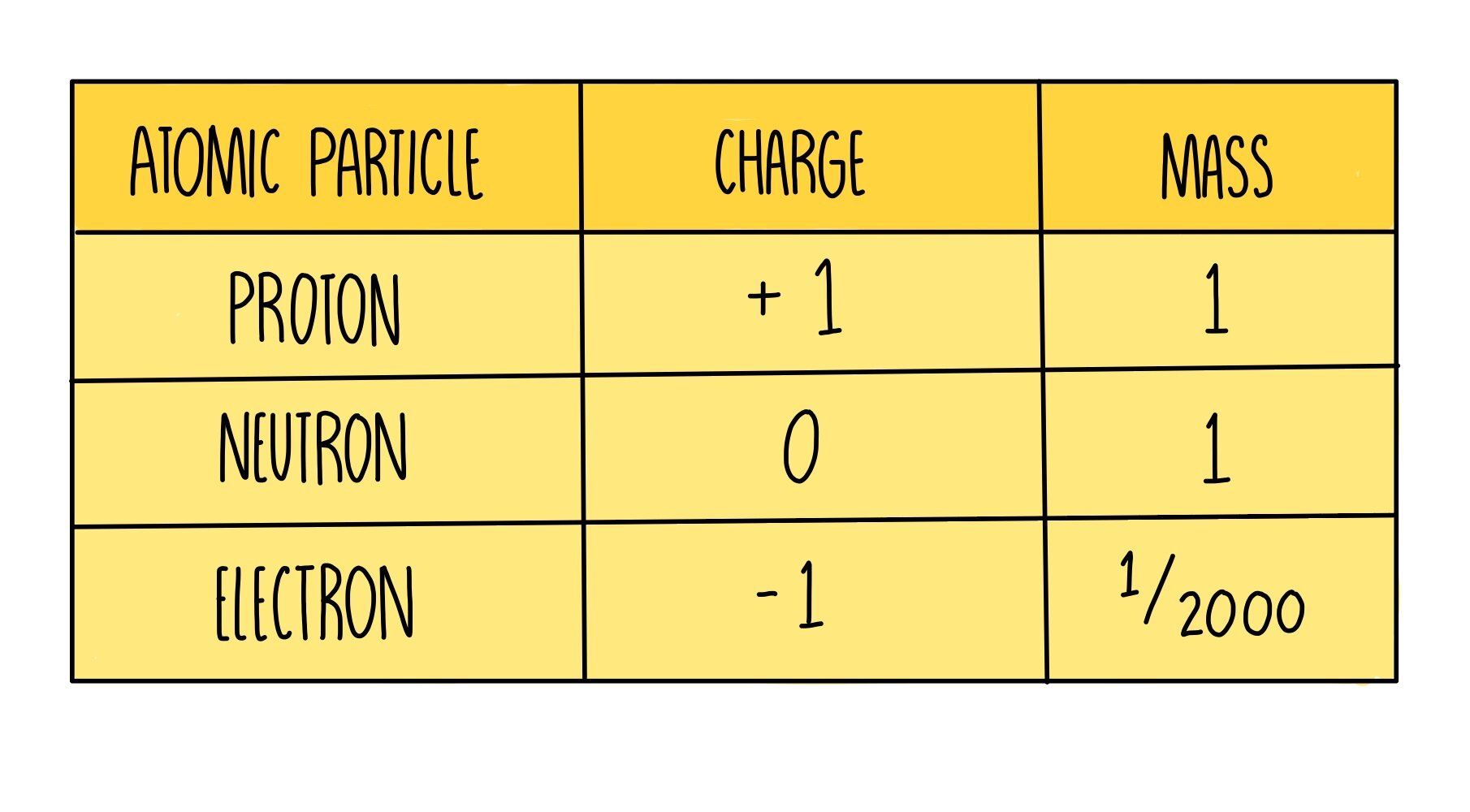
Protons Neutrons And Electrons Chart - Electrons are much smaller than nucleons (protons and neutrons). For example, oxygen atoms have 8 protons, hydrogen atoms only have 1, and gold atoms have 79. A molecule with more protons than electrons will also have a positive charge. Find out the proton definition, along with the mass of a proton and a proton's charge. Do protons have a charge of negative 1? Their number of protons make them a unique type of element. What are protons, electrons and coulombs? If the atom has more electrons than protons, it is called a negative ion. Neutrons, protons, and electrons are the three subatomic particles that compose atoms. Electrons have a negative charge. Do protons have a charge of negative 1? Electrons have a negative charge. Their number of protons make them a unique type of element. A molecule with more protons than electrons will also have a positive charge. Protons :protons are subatomic particles that, together with neutrons, form the nucleus of an atom. For example, oxygen atoms have 8 protons, hydrogen atoms only have 1, and gold atoms have 79. Find out the proton definition, along with the mass of a proton and a proton's charge. Protons are positively charged subatomic particles. This number is like the identity. Neutrons, protons, and electrons are the three subatomic particles that compose atoms. A molecule with more protons than electrons will also have a positive charge. Electrons are much smaller than nucleons (protons and neutrons). Atoms of each element will have a different number of subatomic particles,. What are protons, electrons and coulombs? This number is like the identity. If the atom has more electrons than protons, it is called a negative ion. A molecule with more protons than electrons will also have a positive charge. Protons :protons are subatomic particles that, together with neutrons, form the nucleus of an atom. Their number of protons make them a unique type of element. This number is like the identity. A molecule with more protons than electrons will also have a positive charge. Protons are positively charged subatomic particles. What are protons, electrons and coulombs? Their number of protons make them a unique type of element. Electrons have a negative charge. Protons :protons are subatomic particles that, together with neutrons, form the nucleus of an atom. An atom is made up of neutrons, protons, and electrons. Do protons have a charge of negative 1? Electrons have a negative charge. This number is like the identity. Electrons are much smaller than nucleons (protons and neutrons). Atoms of each element will have a different number of subatomic particles,. An atom is made up of neutrons, protons, and electrons. Outside the nucleus, negatively charged electrons are found orbiting in energy levels. Do protons have a charge of negative 1? Protons are positively charged subatomic particles. Neutrons, protons, and electrons are the three subatomic particles that compose atoms. A molecule with more protons than electrons will also have a positive charge. For example, oxygen atoms have 8 protons, hydrogen atoms only have 1, and gold atoms have 79. Find out the proton definition, along with the mass of a proton. Outside the nucleus, negatively charged electrons are found orbiting in energy levels. Do protons have a charge of negative 1? An atom is made up of neutrons, protons, and electrons. For example, oxygen atoms have 8 protons, hydrogen atoms only have 1, and gold atoms have 79. Electrons have a negative charge. If the atom has more electrons than protons, it is called a negative ion. This number is like the identity. A molecule with more protons than electrons will also have a positive charge. An atom is made up of neutrons, protons, and electrons. Find out the proton definition, along with the mass of a proton and a proton's charge. If the atom has more electrons than protons, it is called a negative ion. Electrons have a negative charge. Protons are positively charged subatomic particles. An atom is made up of neutrons, protons, and electrons. A molecule with more protons than electrons will also have a positive charge. Electrons are much smaller than nucleons (protons and neutrons). Atoms of each element will have a different number of subatomic particles,. If the atom has more electrons than protons, it is called a negative ion. Their number of protons make them a unique type of element. Neutrons, protons, and electrons are the three subatomic particles that compose atoms. This number is like the identity. Find out the proton definition, along with the mass of a proton and a proton's charge. If the atom has more electrons than protons, it is called a negative ion. Outside the nucleus, negatively charged electrons are found orbiting in energy levels. Protons are positively charged subatomic particles. Neutrons, protons, and electrons are the three subatomic particles that compose atoms. An atom is made up of neutrons, protons, and electrons. For example, oxygen atoms have 8 protons, hydrogen atoms only have 1, and gold atoms have 79. Electrons are much smaller than nucleons (protons and neutrons). Electrons have a negative charge. A molecule with more protons than electrons will also have a positive charge. Atoms of each element will have a different number of subatomic particles,. Also, learn where we can find protons in.PPT AQA GCSE Physics 27 Nuclear Physics PowerPoint Presentation, free download ID478811
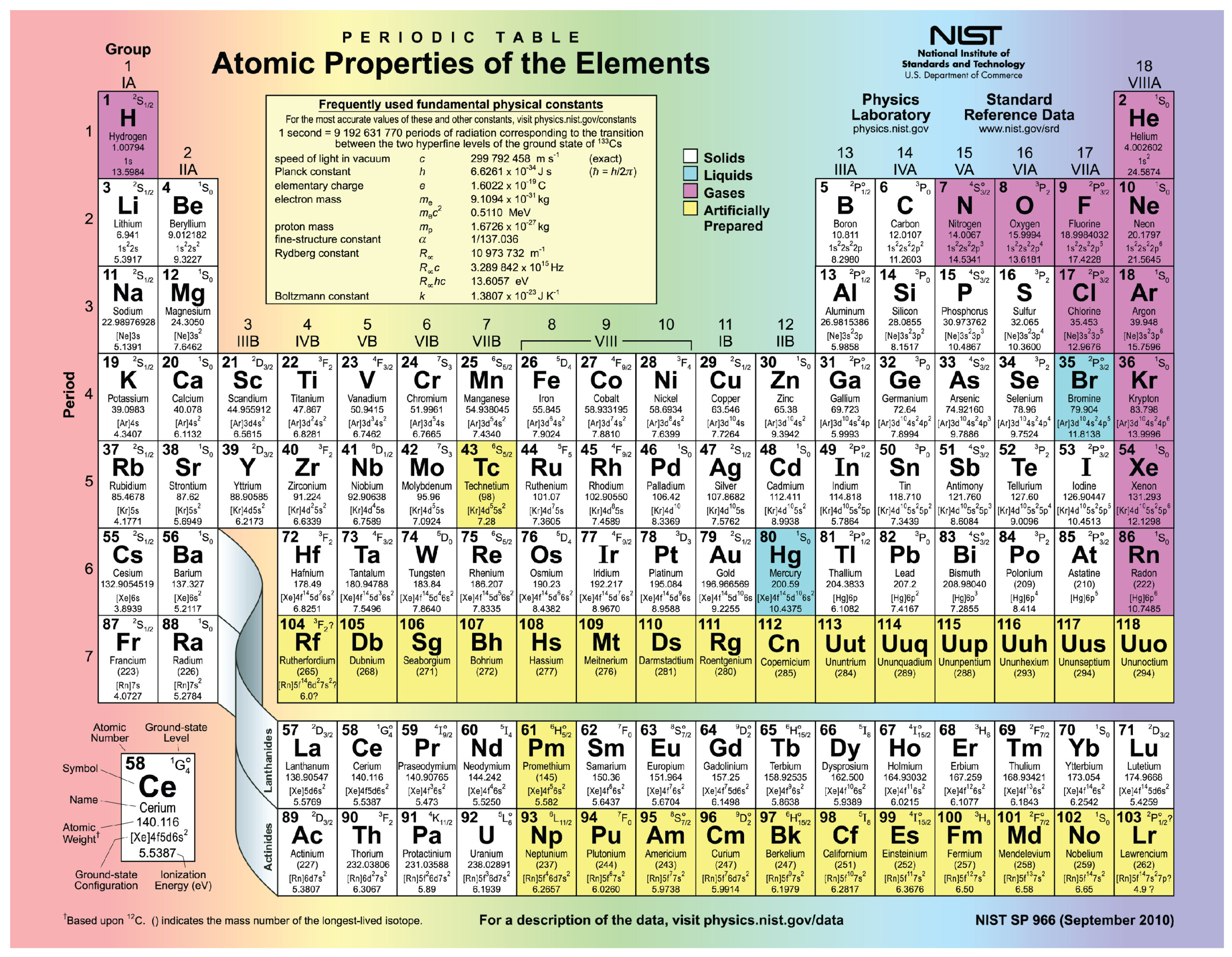
Periodic Table Element Proton Neutron Electron Periodic Table Printable
Neutrons Protons Electrons Periodic Table 2024 Periodic Table Printable
Periodic Table Of Elements List With Protons Neutrons And Electrons Bios Pics
Atomic Structure* — the science sauce
PPT ATOMIC STRUCTURE PowerPoint Presentation, free download ID357852
Periodic table with number of protons neutrons and electrons Brainly.in
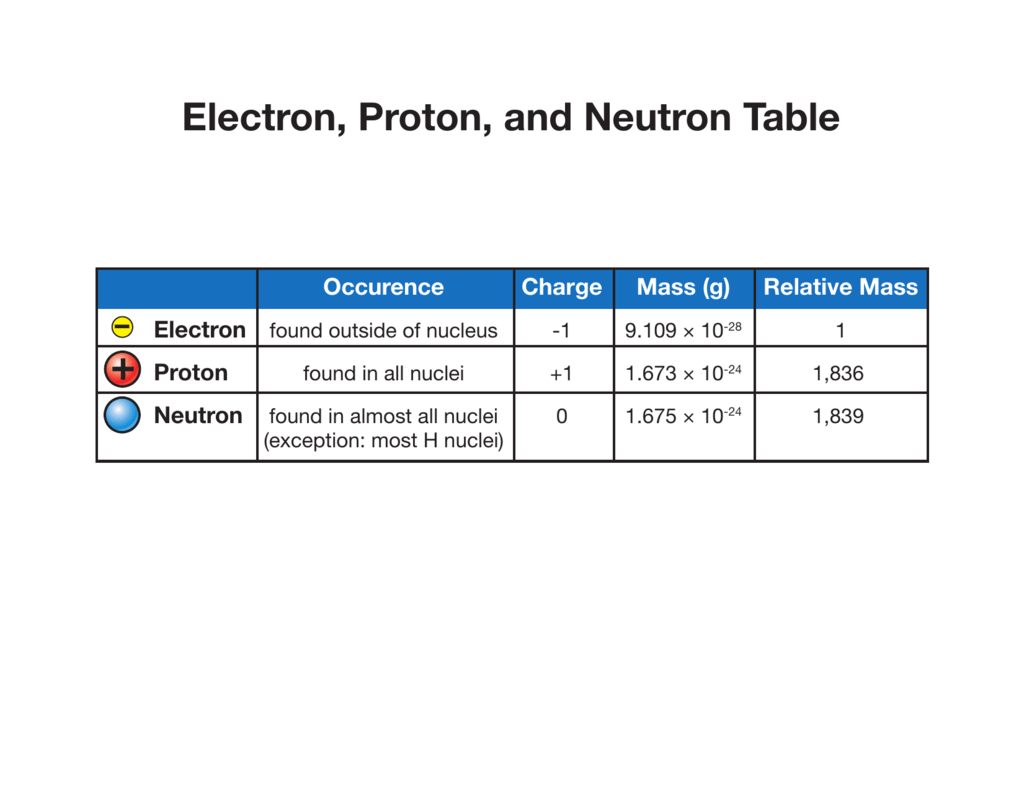
Electron, Proton, and Neutron Table
Periodic Table Of Elements With Protons Neutrons And Electrons
Do Protons Have A Charge Of Negative 1?
Their Number Of Protons Make Them A Unique Type Of Element.
Protons :Protons Are Subatomic Particles That, Together With Neutrons, Form The Nucleus Of An Atom.
What Are Protons, Electrons And Coulombs?
Related Post: