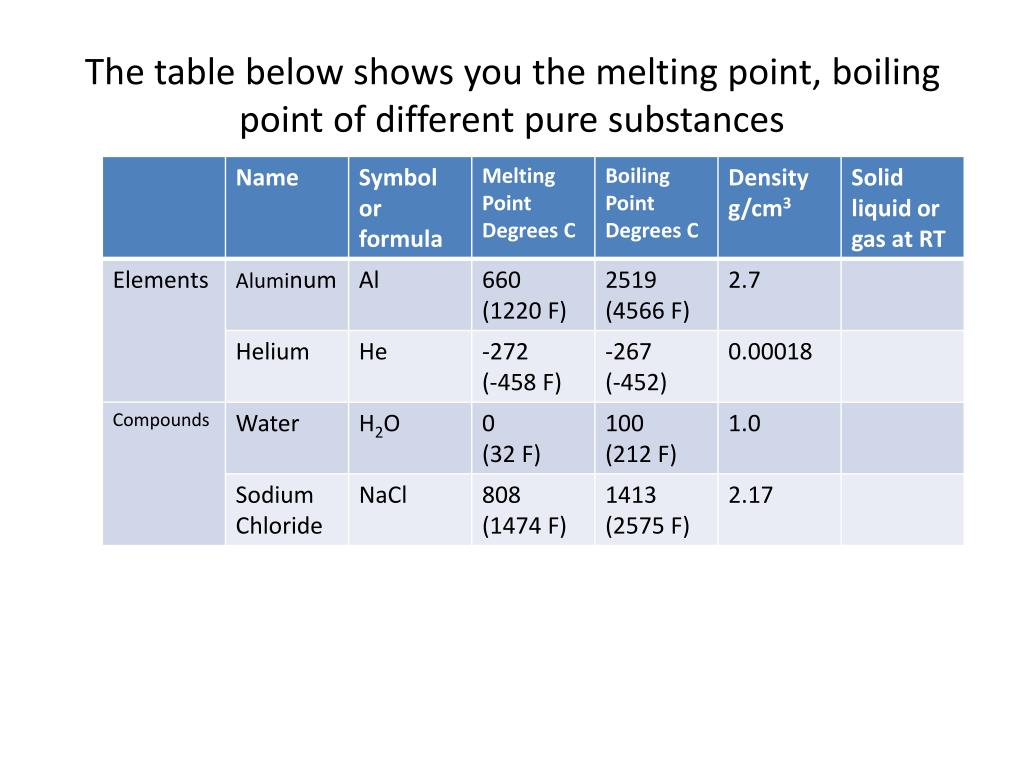
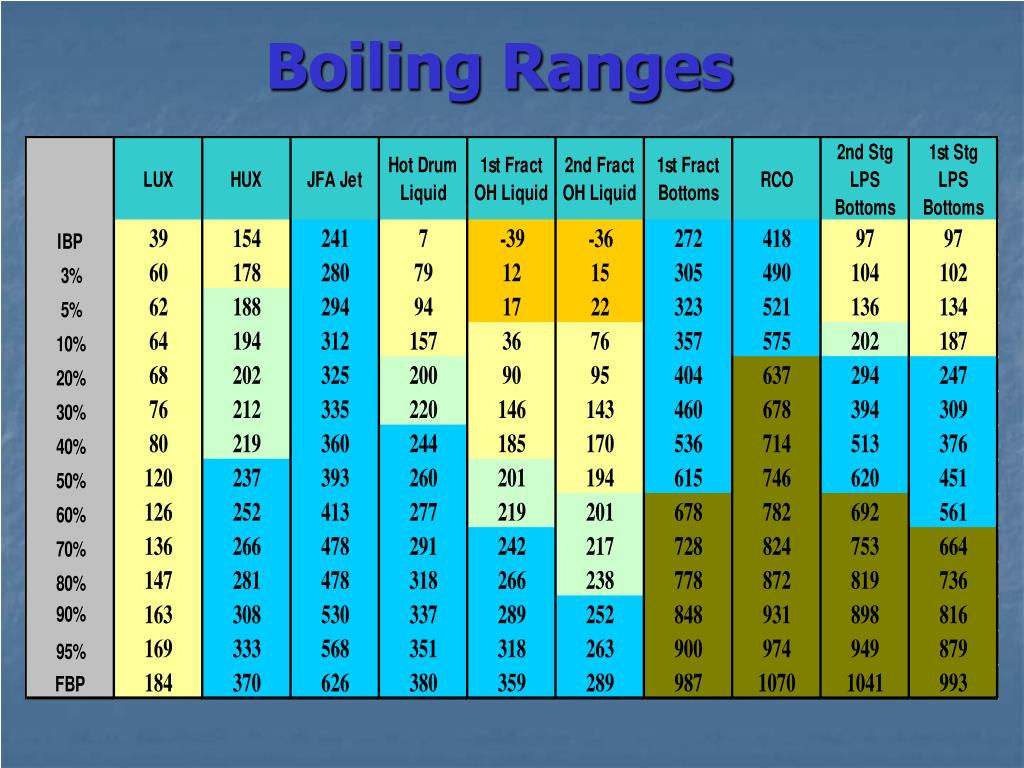
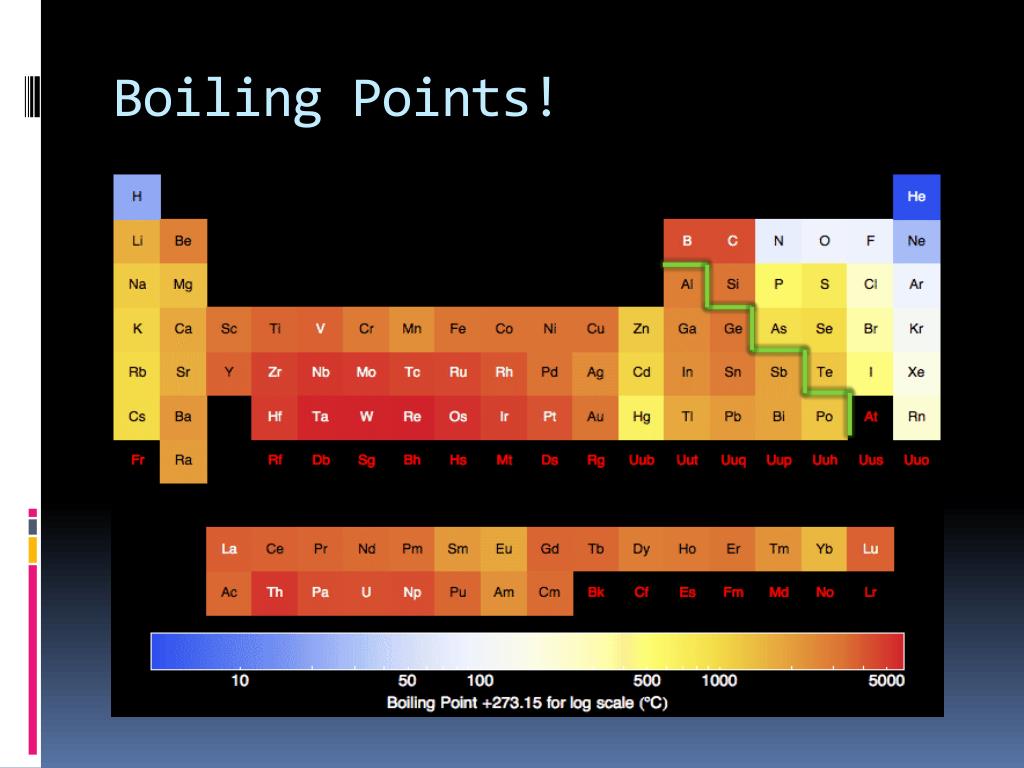
Boiling Point Chart
Boiling Point Chart - This article covers how to boil water for cooking or drinking using your stove, microwave, and more. Boiling occurs when a liquid is heated to its boiling point, so that the vapour. Learn about the boiling point of water. Boiling occurs when a liquid reaches a. See what temperature water boils at in celsius, fahrenheit, and kelvin. The boiling point of a substance is the temperature at which the vapor pressure of a liquid equals the pressure surrounding the liquid [1][2] and the liquid changes into a vapor. The meaning of boiling is heated to the boiling point. The lower the pressure of a gas above a liquid, the lower the temperature at. If so, you may need to boil water. Boiling is a physical process in which a liquid changes into a gas. This transition from one state to another is called a phase change or phase transition. Boiling point, temperature at which the pressure exerted by the surroundings upon a liquid is equaled by the pressure exerted by the vapor of the liquid; This occurs when water molecules gain. The boiling point of a substance is the temperature at which the vapor pressure of a liquid equals the pressure surrounding the liquid [1][2] and the liquid changes into a vapor. Boiling a liquid boils at a temperature at which its vapor pressure is equal to the pressure of the gas above it. At its core, boiling represents a phase transition from the liquid state to the gaseous state, more commonly known as vapor. Boiling is a physical process in which a liquid changes into a gas. See what temperature water boils at in celsius, fahrenheit, and kelvin. How to use boiling in a sentence. Boiling occurs when a liquid reaches a. How to use boiling in a sentence. Boiling point, temperature at which the pressure exerted by the surroundings upon a liquid is equaled by the pressure exerted by the vapor of the liquid; The reverse of boiling is condensation. This transition from one state to another is called a phase change or phase transition. Boiling a liquid boils at a. At its core, boiling represents a phase transition from the liquid state to the gaseous state, more commonly known as vapor. This article covers how to boil water for cooking or drinking using your stove, microwave, and more. The reverse of boiling is condensation. This transition from one state to another is called a phase change or phase transition. If. Boiling a liquid boils at a temperature at which its vapor pressure is equal to the pressure of the gas above it. Boiling or ebullition is the rapid phase transition from liquid to gas or vapour; Boiling occurs when a liquid reaches a. This occurs when water molecules gain. The reverse of boiling is condensation. If so, you may need to boil water. Boiling a liquid boils at a temperature at which its vapor pressure is equal to the pressure of the gas above it. Boiling or ebullition is the rapid phase transition from liquid to gas or vapour; This occurs when water molecules gain. Boiling occurs when a liquid reaches a. See what temperature water boils at in celsius, fahrenheit, and kelvin. This article covers how to boil water for cooking or drinking using your stove, microwave, and more. At its core, boiling represents a phase transition from the liquid state to the gaseous state, more commonly known as vapor. Boiling occurs when a liquid is heated to its boiling point,. The lower the pressure of a gas above a liquid, the lower the temperature at. Boiling a liquid boils at a temperature at which its vapor pressure is equal to the pressure of the gas above it. This transition from one state to another is called a phase change or phase transition. The meaning of boiling is heated to the. If so, you may need to boil water. Boiling point, temperature at which the pressure exerted by the surroundings upon a liquid is equaled by the pressure exerted by the vapor of the liquid; Boiling or ebullition is the rapid phase transition from liquid to gas or vapour; Boiling occurs when a liquid is heated to its boiling point, so. Boiling is a physical process in which a liquid changes into a gas. See what temperature water boils at in celsius, fahrenheit, and kelvin. How to use boiling in a sentence. Boiling a liquid boils at a temperature at which its vapor pressure is equal to the pressure of the gas above it. If so, you may need to boil. Boiling or ebullition is the rapid phase transition from liquid to gas or vapour; This transition from one state to another is called a phase change or phase transition. The meaning of boiling is heated to the boiling point. Boiling a liquid boils at a temperature at which its vapor pressure is equal to the pressure of the gas above. Learn about the boiling point of water. Boiling or ebullition is the rapid phase transition from liquid to gas or vapour; The lower the pressure of a gas above a liquid, the lower the temperature at. Boiling is a physical process in which a liquid changes into a gas. This transition from one state to another is called a phase. At its core, boiling represents a phase transition from the liquid state to the gaseous state, more commonly known as vapor. Boiling occurs when a liquid is heated to its boiling point, so that the vapour. Boiling is the process by which a liquid turns into a vapor when it is heated to its boiling point. Boiling occurs when a liquid reaches a. The boiling point of a substance is the temperature at which the vapor pressure of a liquid equals the pressure surrounding the liquid [1][2] and the liquid changes into a vapor. Boiling is a physical process in which a liquid changes into a gas. The change from a liquid phase to a gaseous phase occurs when the vapor pressure of the liquid. If so, you may need to boil water. The reverse of boiling is condensation. Boiling a liquid boils at a temperature at which its vapor pressure is equal to the pressure of the gas above it. This transition from one state to another is called a phase change or phase transition. See what temperature water boils at in celsius, fahrenheit, and kelvin. How to use boiling in a sentence. Learn about the boiling point of water. The lower the pressure of a gas above a liquid, the lower the temperature at. Boiling or ebullition is the rapid phase transition from liquid to gas or vapour;Melting Points And Boiling Points
Engine Coolant Boiling Point Chart
PPT Organic Chemistry PowerPoint Presentation, free download ID1810137
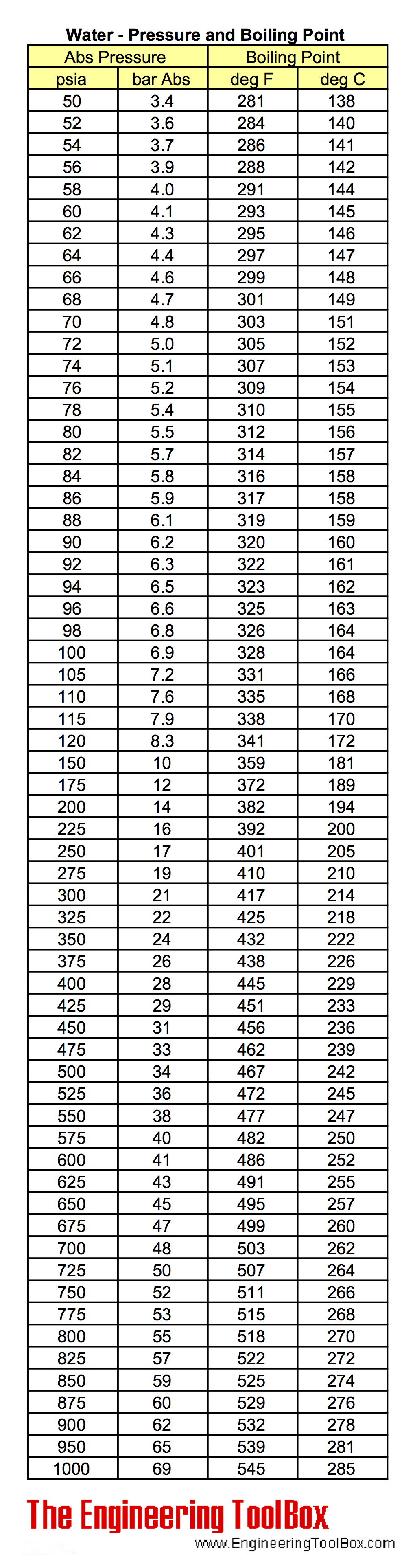
Water Pressure and Boiling Points
Coolant Boiling Point Chart at Christine Hibbler blog
C2cl4 Boiling Point
Melting And Boiling Point Of Water In Kelvin at Angelina Otto blog
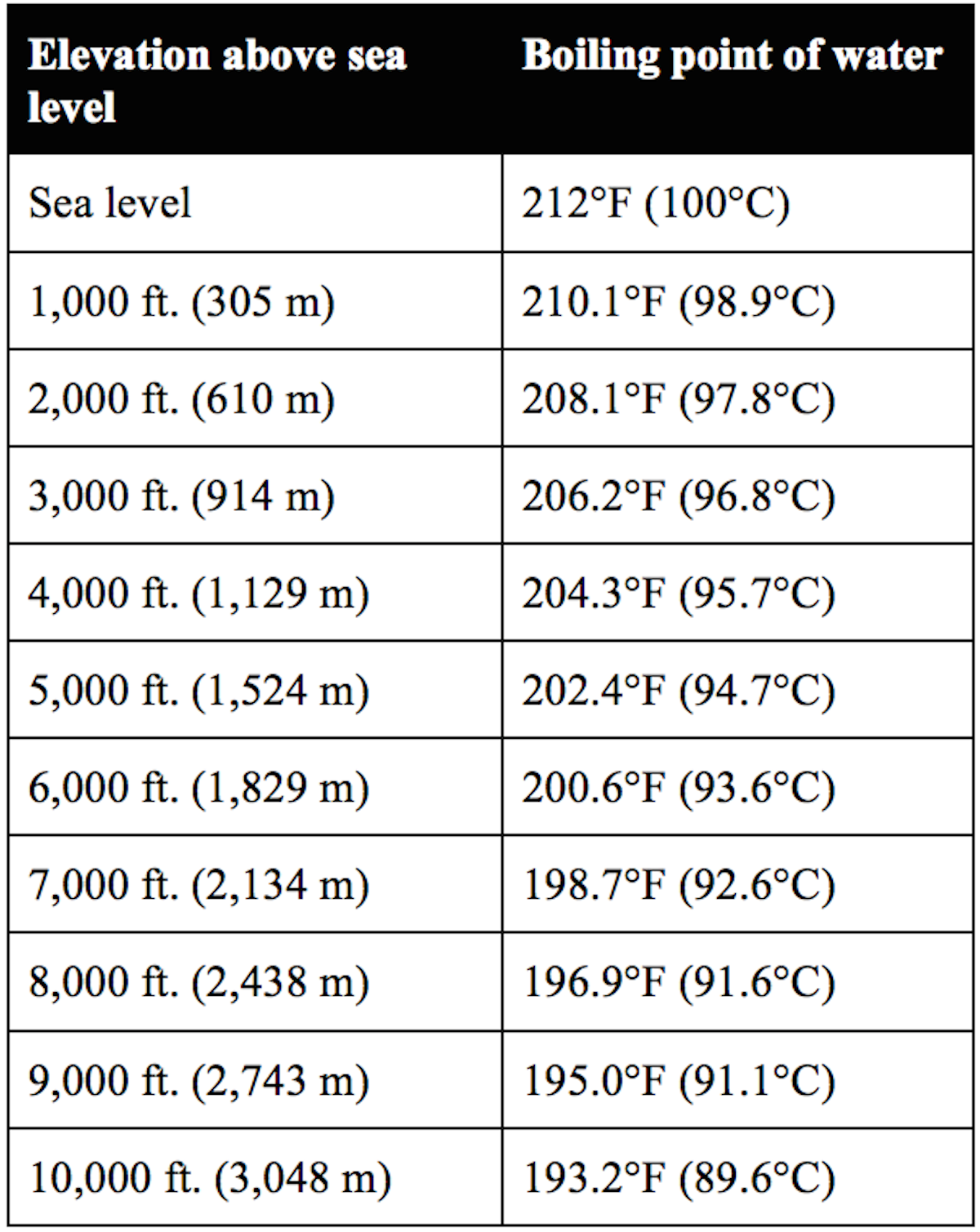
boiling point altitude chart Keski
Chlorine Boiling Point
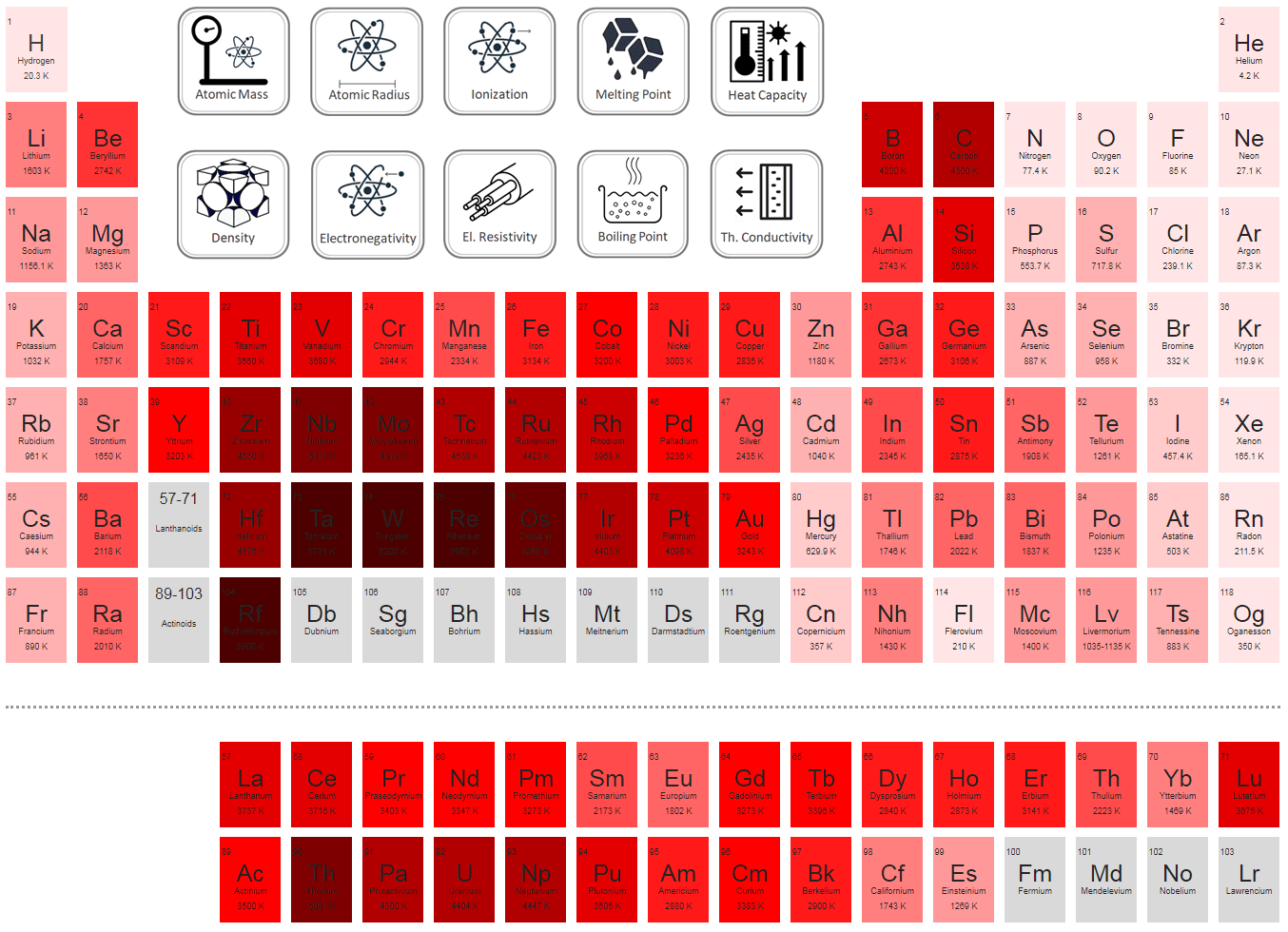
Bulk Properties of Water Presentation Chemistry
This Occurs When Water Molecules Gain.
This Article Covers How To Boil Water For Cooking Or Drinking Using Your Stove, Microwave, And More.
Boiling Point, Temperature At Which The Pressure Exerted By The Surroundings Upon A Liquid Is Equaled By The Pressure Exerted By The Vapor Of The Liquid;
The Meaning Of Boiling Is Heated To The Boiling Point.
Related Post:

.PNG)

.PNG)